New Method Uses Light and Gold Nanoparticles for Highly Targeted, Non-Invasive Drug Delivery
Researchers in the Technion Faculty of Biotechnology and Food Engineering have developed a new drug-release method that uses light for highly targeted, completely non-invasive delivery
HAIFA, ISRAEL (February 25, 2018) – Over the last century, there has been astounding progress in medical science, leading to the development of efficient, effective medications for treating cancer and a wide variety of other diseases. But the random dispersion of drugs throughout the body often lowers their effectiveness and, even worse, damages healthy tissue. A prime example of this is the use of chemotherapy drugs, which work to block cell division, causing hair loss and bowel issues in cancer patients (hair growth and waste elimination both depend on rapid cell turnover).
This has led to a global effort to develop smarter systems for drug delivery that will more effectively target the specific part of the body affected by cancer, bypassing healthy tissue. A recent issue of ACS Applied Materials & Interfaces presents groundbreaking work in the field by the Technion Faculty of Biotechnology and Food Engineering.
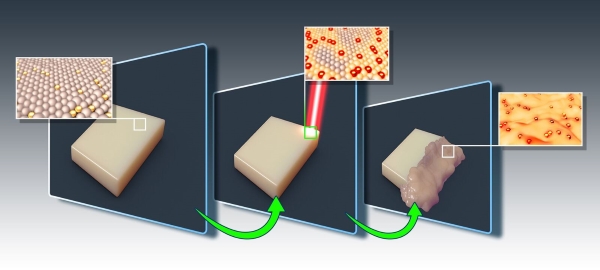

Doctoral candidate Alona Shagan and Assistant Professor Boaz Mizrahi have developed a technology that enables drugs to be delivered and released only to the diseased tissue which the drug is targeting. The new method uses a unique polymer coating that contains nanoscale gold particles, in addition to the drug itself. The drug only releases when a light shines on the gold particles, causing the polymeric coating to melt.

“Photo-triggered materials fulfill a vital role in a range of bio-medical applications,” said Shagan. “But despite this enormous potential, these materials are rarely used because of toxins in the polymer coating itself, and damage caused by high-energy (shortwave) light.”
The researchers designed the one-of-a-kind delivery method to release under longwave light (Near-Infrared, NIR). The light warms the gold nanoshells, melting the polymer packaging, and releasing the drug. The primary advantage of NIR light is its ability to penetrate bodily tissues without harming them.
“We’ve developed a material with varying melting points, allowing us to control it using low intensities,” explains Prof. Mizrahi. “Our system is composed of FDA-approved materials, and we are relatively close to clinical application.”
The researchers believe this new technology can be used for a variety of other applications, such as sealing of internal and external injuries, temporary holding of tissue during surgery, or as biodegradable scaffolds for growing transplant organs. It may even be possible to use the polymer as part of the self-healing process, giving it a wide range of both medical and non-medical applications.
“This article focuses on the concept and material: how we can design the material to fulfill these particular physical and mechanical requirements,” says Prof. Mizrahi. “The next step will include creating particles that include the drugs so that we can test their improved effectiveness using this delivery technology. We’ll discuss that in an upcoming article.”
