To celebrate the decade of Nobel Prizes, Technion has released an elite selection of silk Nobel scarves and ties.

A Decade of Nobel Prizes
(Writen on 10.12.2014)
Ten years ago, on December 10th, 2004, Technion became home to Israel’s 1st Nobel Prizes in the natural sciences. Since then the Technion Nobel legacy has continued, with the 2011 Nobel Prize in Chemistry awarded to Distinguished Prof. Dan Shechtman, and the 2013 Prize in Chemistry awarded to Technion graduate Prof. Arieh Warshel.
Ubiquitin: so called, because it is a protein present in all living cells. No-one knew why it was there, and no-one dared to wonder: it was just boring – “ubiquitous”.
But no living secrets are untouched by Technion scientists.
Throughout the ‘70s and ‘80s, Distinguished Professors Avram Hershko and Aaron Ciechanover unveiled the mysteries of the ubiquitin system, revealing some masterkeys of human health. The ubiquitous protein ubiquitin, they showed, is the key factor in deciding when and how a cell should regenerate. Imbalance in ubiquitin reveals itself in some of the world’s most incurable afflictions – such as cancer and neuro-degenerative disorders.
By 2004, the Technion research was already revolutionizing medical understanding and opening the way to innovative cures and treatments. No wonder that, in that year, the two Technion Professors became Israel’s first Nobel Laureates in science.
The Ubiquitin Story
In 1975, a protein of unknown function was identified by Dr. Gideon Goldstein which he called Ubiquitin as he thought it was probably ubiquitous to all living cells – turning up everywhere in animal and plant cells and even yeast. Each living cell is made up of many tiny proteins. A protein is a molecule made up of one or more chains of amino acids in a predetermined order. Proteins maintain structure, function and regulation of cells. Each protein has its own unique function – some famous proteins being hormones, enzymes and antibodies.
In the 1970s, protein synthesis was understood, yet the breakdown or destruction of unwanted proteins in cells back to amino acid was still quite a mysterious process.
Over three decades ago, Technion Profs. Hershko and Ciechanover were immersed in ideas as to how to complete our picture of cell regeneration through understanding how proteins are degraded. Working closely with colleague and fellow Nobel Prize Laureate Irwin Rose, then of Fox Chase Cancer Center, Philadelphia, they showed how the small and common protein Ubiquitin attaches to other proteins, and marks them for destruction. Ubiquitin is quite a unique protein, as its task is that of a kind of runner between other proteins, labeling them if necessary for destruction or degradation. “Many knew how the body produces proteins, but not how they were destroyed,” says Hershko. “Without an engine, a car cannot run; without brakes, it is out of control. Proteins provide ways to moderate the body’s machinery.”
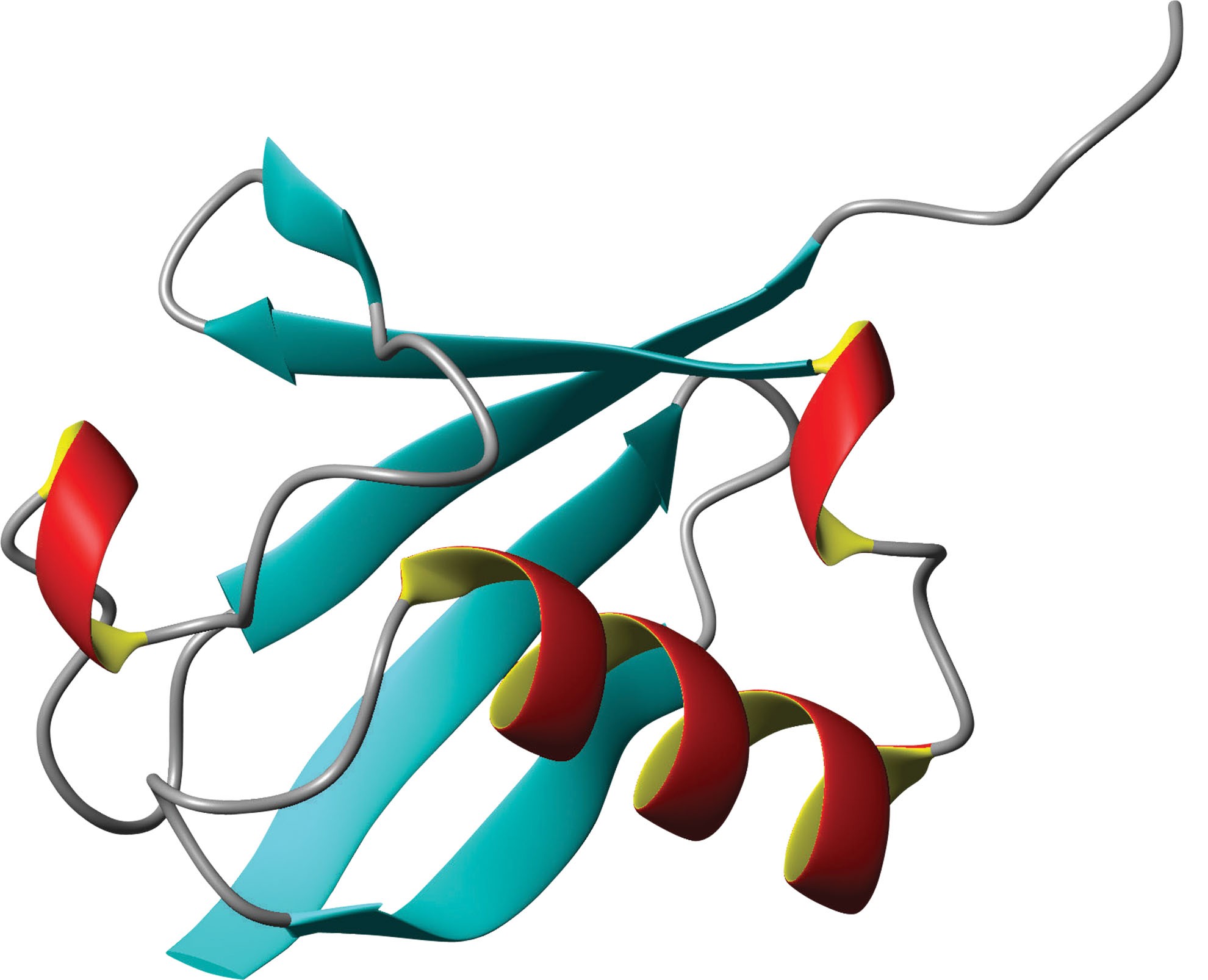
At first the three scientists, Hershko, graduate student Ciechanover and U.S. colleague Irwin Rose noticed that Ubiqitin had a way of binding to other proteins – but they didn’t know why. “Hershko used a really simple system in order to make the discovery – just a soup of enzymes and proteins,” colleague John Mayer of Nottingham University told New Scientist. “From this he was able to show the target protein must be “ubiquitinated.”
Sometimes working in tandem, Ubiquitin molecules smartly seek out proteins that are no longer needed, damaged or unhelpful and tag them for degradation, escorting them to a barrel-shaped structure called a proteasome – the cellular recycler. It is a process that cognoscenti call “ubiquitination”. It is a “kiss of death” for protein, which is a “kiss of life” for cells. Later, the scientists identified three types of enzymes involved in the ubiquitination process. The third type – the Ubiquitin protein ligases – is the one that identifies and singles out the target protein. Ligases are the cellular whistle-blowers. The sophisticated process takes place in cells all over the body – it is highly ubiquitous. But at first, few attached the label of “tremendous discovery” to the work.

